Table of Contents
Does HCN have a linear shape?
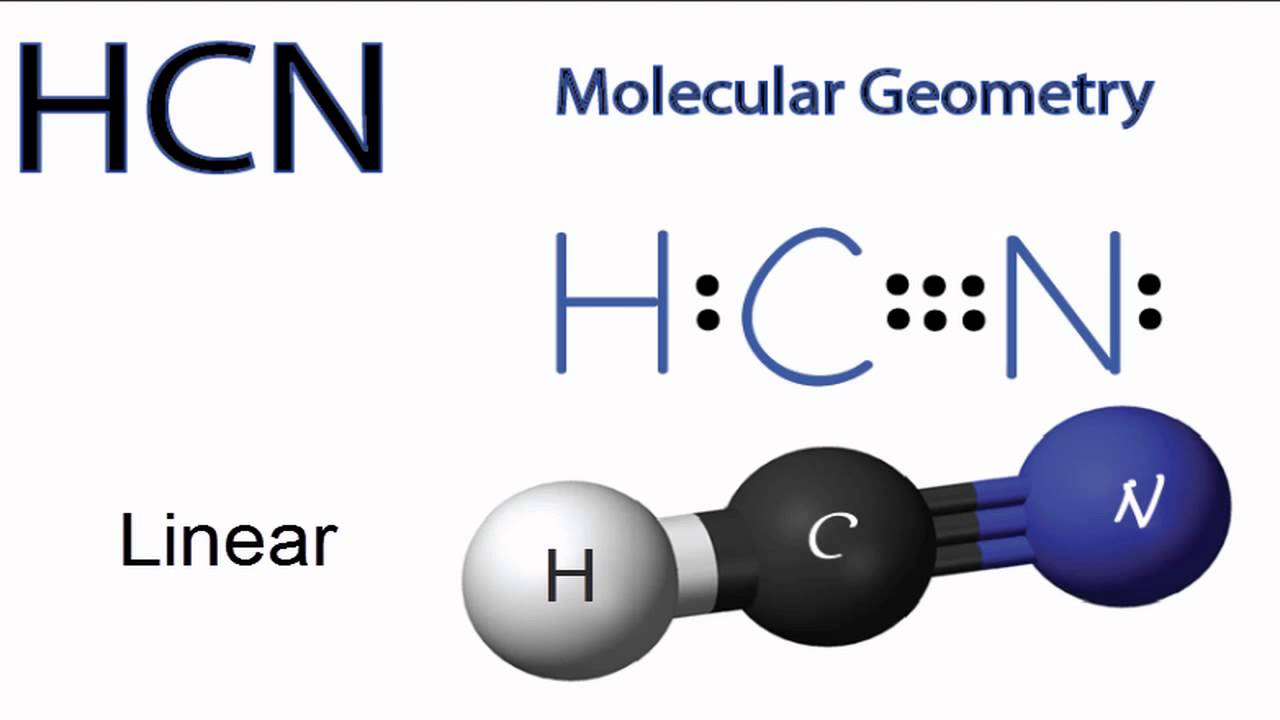
HCN has a total of 10 valence electrons. It is covered under AX2 molecular geometry and has a linear shape. The bond angles of HCN is 180 degrees.
Is HCN linear or tetrahedral?
HCN has two electron-dense areas around the central atom; therefore, it is linear in shape.
Is HCN linear or trigonal planar?
linear
What is the shape of HCN?
linear
Is HCN Lewis structure linear?
HCN has two electron-dense areas around the central atom; therefore, it is linear in shape.
Is HCN linear Vsepr?
In HCN lewis structure, carbon forms one single bond with the hydrogen atom and a triple bond with the nitrogen atom. The bond angle is 180 degrees and there are 10 valence electrons. HCN is a polar molecule with linear geometry
Is HCN a tetrahedral?
HCN only has two electron-dense areas around the central atom; therefore, it cannot be tetrahedral in shape.
Is HCN linear?
HCN, hydrogen cyanide, is a volatile and poisnous compound with distinguished bitter odor. It is linear molecule with a triple bond between C and N atom and has bond angle of 180 degrees.
What is the molecular shape of HCN?
linear
Is HCN a trigonal planar?
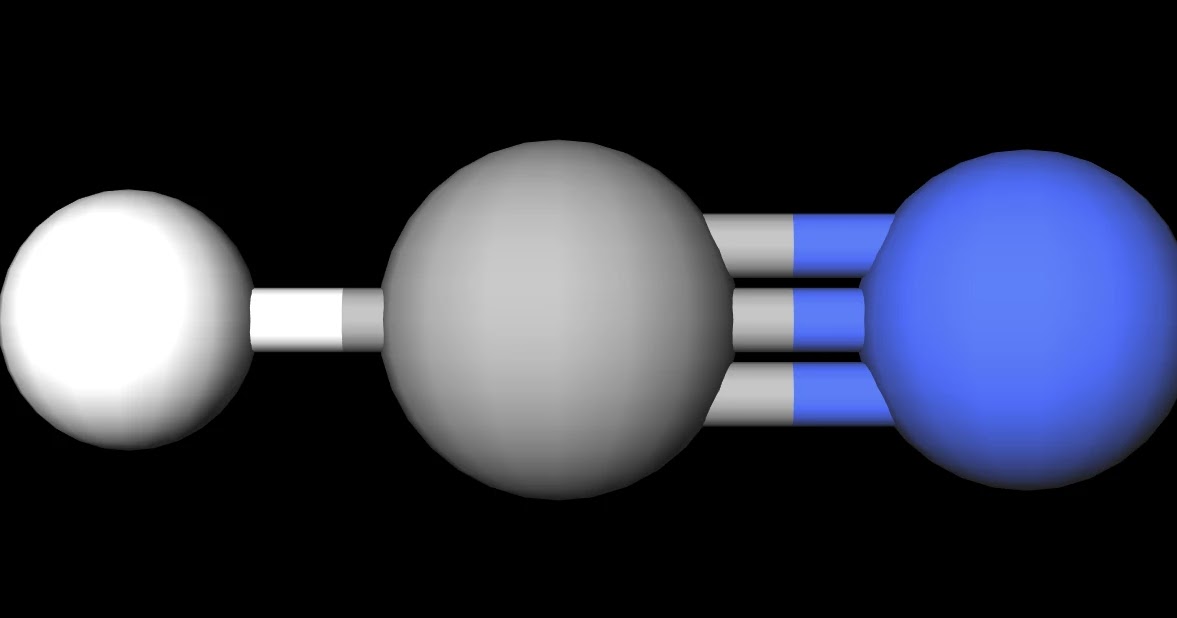
HCN only has two electron-dense areas around the central atom; therefore, it cannot be trigonal planar in shape.
Is HCN a linear shape?
HCN has a total of 10 valence electrons. It is covered under AX2 molecular geometry and has a linear shape. The bond angles of HCN is 180 degrees.